Evaluation of the anti-inflammatory and antioxidant activities of algae-mediated zinc oxide nanoparticles
DOI:
https://doi.org/10.26832/24566632.2025.100202Keywords:
Algae, BSA, Bioactive agents, Green synthesis, Zinc oxide nanoparticlesAbstract
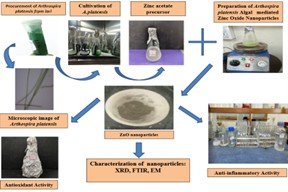
The current study investigates the anti-inflammatory and antioxidant potential of zinc oxide (ZnO) nanoparticles synthesized using an algal extract as a green, eco-friendly reducing and stabilizing agent. The biosynthesized nanoparticles were characterized using UV-Vis spectroscopy (UV, absorption peak 330 nm) XRD (24 nm crystalline size), SEM (rod shaped morphology) and FTIR (Zn-O bond at 671cm-1). Anti-inflammatory activity was assessed using the bovine serum albumin (BSA) protein denaturation method showing dose dependent inhibition (28.4-88.0 % at 100- 500 ug/ml; IC50 = 251.64 ug/ml), comparable to ibuprofen (IC50 = 233.60 ug/ml). Antioxidant activity of green synthesised ZnO nanoparticles was evaluated using DPPH free radical scavenging assay (28.8-88.0% at 100- 500 ug/ml; IC50 = 200.06 ug/ml) attaining ~ 85% efficacy of ascorbic acid (IC50 = 171ug/ml). Results showed that ZnO NPs exhibited strong, concentration-dependent anti-inflammatory activity, inhibiting BSA denaturation by 28.4% to 88% over a range of 100–500 µg/mL, with an IC50 value of 251.64 µg/mL, closely comparable that of ibuprofen (IC50=233.6 µg/mL). Moreover, the nanoparticles showed substantial antioxidant activity, with DPPH radical scavenging ranging from 28.8% to 88.9% within the same concentration range, and an IC50 of 200.06 µg/mL—comparable to ascorbic acid (IC50=171 µg/mL). The bioactivities are likely enhanced by the phytochemicals present in A. platensis, such as phenolic compounds and C-phycocyanin. Thus, algae-mediated ZnO nanoparticles exhibit significant anti-inflammatory and antioxidant effects in a dose-dependent manner, suggesting their potential as bioactive agents for pharmaceutical and therapeutic applications.
Downloads
References
Ahmed, N. A., & Othman, A. S. (2024). Green fabrication of ZnO nanoparticles via Spirulina platensis and its efficiency against biofilm forming pathogens. Microbial Cell Factories, 23(1), 92. https://doi.org/10.1186/s12934-024-02360-x
Balls, M. (2022). Alternatives to laboratory animals: Trends in replacement and the three rs. Alternatives to Laboratory Animals, 50(1), 10-26. https://doi.org/10.1177/02611929221082250
Behra, R., Sigg, L., Clift, M. J., Herzog, F., Minghetti, M., Johnston, B., Petri-Fink, A., & Rothen-Rutishauser, B. (2013). Bioavailability of silver nanoparticles and ions: from a chemical and biochemical perspective. Journal of the Royal Society Interface, 10(87), 20130396. https://doi.org/10.1098/rsif.2013.0396
Blois, M.S. (1958). Antioxidant determinations by the use of a stable free radical. Nature, 181(4617), 1199-1200. https://doi.org/10.1038/1811199a0
Curran, A. M., Girgis, A. A., Jang, Y., Crawford, J. D., Thomas, M. A., Kawalerski, R., Coller, J., Bingham III, C.O., Na, C.H., & Darrah, E. (2023). Citrullination modulates antigen processing and presentation by revealing cryptic epitopes in rheumatoid arthritis. Nature communications, 14(1), 1061. https://doi.org/10.1038/s41467-023-36620-y
Gunathilake, K. D. P. P., Ranaweera, K. K. D. S., & Rupasinghe, H. V. (2018). In vitro anti-inflammatory properties of selected green leafy vegetables. Biomedicines, 6(4), 107. https://doi.org/10.3390/biomedicines6040107
Harris, J., Malaiappan, S., & Shanmugam, R. (2023). The development and evaluation of melatonin-loaded, calcium oxide nanoparticle-based neem and clove extract: an in vitro study. Cureus, 15(9). https://doi.org/10.7759/cureus.46293
Kanika, Durgapal, M., Arachne, & Kumar, M. (2025). Green synthesis and antimicrobial activities of Arthospira platensis zinc oxide nanoparticles. Environment Conservation Journal. https://doi.org/10.36953/ECJ.30652998
Knight, A. (2011). The costs and benefits of animal experimentation. In The Costs and Benefits of Animal Experiments (pp. 179-193). London: Palgrave Macmillan UK. https://doi.org/10.1057/9780230306417_12
Leelaprakash, G., & Dass, S. M. (2011). Invitro anti-inflammatory activity of methanol extract of Enicostemma axillare. International Journal of Drug Development and Research, 3(3), 189-196. http://www.ijddr.in
Lopez-Miranda, J. L., Molina, G. A., González-Reyna, M. A., España-Sánchez, B. L., Esparza, R., Silva, R., & Estévez, M. (2023). Antibacterial and anti-inflammatory properties of ZnO nanoparticles synthesized by a green method using Sargassum extracts. International Journal of Molecular Sciences, 24(2), 1474. https://doi.org/10.3390/ijms24021474
Nagajyothi, P. C., Cha, S. J., Yang, I. J., Sreekanth, T. V. M., Kim, K. J., & Shin, H. M. (2015). Antioxidant and anti-inflammatory activities of zinc oxide nanoparticles synthesized using Polygala tenuifolia root extract. Journal of Photochemistry and Photobiology B: Biology, 146, 10-17. https://doi.org/10.1016/j.jphotobiol.2015.02.008
Nandhini, J., Karthikeyan, E., & Rajeshkumar, S. (2024). Green synthesis of zinc oxide nanoparticles: eco-friendly advancements for biomedical marvels. Resources Chemicals and Materials. 3(4), 294-316. https://doi.org/10.1016/j.recm.2024.05.001
Pati, R., Das, I., Mehta, R. K., Sahu, R., & Sonawane, A. (2016). Zinc-oxide nanoparticles exhibit genotoxic, clastogenic, cytotoxic and actin depolymerization effects by inducing oxidative stress responses in macrophages and adult mice. Toxicological Sciences, 150(2), 454-47. https://doi.org/10.1093/toxsci/kfw010
Rajeshkumar, S., Santhoshkumar, J., Kumar, P. S., Saravanan, A., & Tharani, M. (2023). Characterization and evaluation of cytotoxic effect, antioxidant and antimicrobial activities of zinc oxide nanoparticles derived from Justicia adhatoda. Applied Nanoscience, 13(6), 3993-4004. https://doi.org/10.1007/s13204-022-02670-9
Ramesh, M., Anbuvannan, M., & Viruthagiri, G. J. S. A. P. A. M. (2015). Green synthesis of ZnO nanoparticles using Solanum nigrum leaf extract and their antibacterial activity. Spectrochimica Acta Part A: Molecular and Biomolecular Spectroscopy, 136, 864-870. https://doi.org/10.1016/j.saa.2014.09.105
Ravipati, A. S., Zhang, L., Koyyalamudi, S. R., Jeong, S. C., Reddy, N., Bartlett, J., Smith, P.T., Shanmugam, K., Münch, G., Wu, M.J., & Satyanarayanan, M. (2012). Antioxidant and anti-inflammatory activities of selected Chinese medicinal plants and their relation with antioxidant content. BMC Complementary and Alternative Medicine, 12, 1-14. http://www.biomedcentral.com/1472-6882/12/173
Varghese, R. M., Kumar, A., & Shanmugam, R. (2024). Comparative anti-inflammatory activity of silver and zinc oxide nanoparticles synthesized using Ocimum tenuiflorum and Ocimum gratissimum herbal formulations. Cureus, 16(1). https://doi.org/10.7759/cureus.53562
Williams, L. A. D., O'connar, A., Latore, L., Dennis, O., Ringer, S., Whittaker, J. A., Conrad, J., Vogler, B., & Rosner, H. (2008). The in vitro anti-denaturation effects induced by natural products and non-steroidal compounds in heat treated (immunogenic) bovine serum albumin is proposed as a screening assay for the detection of anti-inflammatory compounds, without the use of animals. West Indian Medical Journal, 57(4), 327-331. https://www.mona.uwi.edu/fms/wimj/article/764
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Agriculture and Environmental Science Academy

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.




