Environmental factors affecting Trichoderma spp. and their biocontrol potential in post-harvest disease management
DOI:
https://doi.org/10.26832/24566632.2025.1002014Keywords:
Culture media, pH, Photoperiodicity, Salinity, TemperatureAbstract
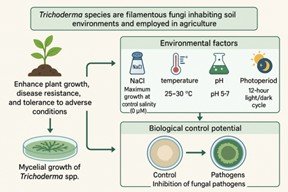
Trichoderma species are filamentous fungi inhabiting soil environments and employed in agriculture due to their capacity to enhance plant growth, disease resistance, and tolerance to adverse environmental conditions. This research aimed to examine the influence of NaCl concentration, temperature, pH, and photoperiodicity on the growth of four Trichoderma species, and to evaluate the potential of Trichoderma spp. as a biological control agent against five post-harvest fungal pathogens under laboratory conditions. Mycelial growth of Trichoderma species exhibited a negative correlation with salinity. Maximum growth rates (4.35-4.59 cm) were observed at control salinity (0 µM). Potato Dextrose Agar media consistently supported significantly (p ≤ 0.05) higher mycelial growth (4.33-4.35 cm) than Malt Extract Agar, Carrot Agar, and Komada for all Trichoderma species. The optimal temperature for mycelial growth of Trichoderma spp. ranged from 25-30°C, with maximum growth rates of 4.12-4.40 cm. Temperatures below 20°C and above 35°C resulted in substantial growth reduction, demonstrating temperature's critical influence on mycelial development. Besides, the optimal pH for mycelial growth of Trichoderma spp. ranged from 5 to 7, with maximum growth rates of 4.12-4.43 cm. Mycelial growth of Trichoderma spp. was significantly enhanced under a 12-hour light/dark cycle (4.33-4.43 cm) compared to continuous light (2.24-2.79 cm) and continuous darkness (2.28-2.56 cm) conditions. Therefore, Trichoderma spp. treatments significantly inhibited the mycelial growth of five fungal pathogens compared to the control group, among them, T. koningii demonstrated the highest level of inhibition, ranging from 0.28 cm (B. cinerea) to 1.65 cm (A. niger).
Downloads
References
Abu-Shanab, N. S., Alananbeh, K. M., Othman, Y. A., & Al-Ajlouni, M. G. (2022). Effect of using Trichoderma spp. on turfgrass quality under different levels of salinity. Water, 14,3943. https://doi.org/10.3390/w14233943
Alwadai A.S., Perveen, K., & Alwahaibi M. (2022). The isolation and characterization of antagonist Trichoderma spp. from the Soil of Abha, Saudi Arabia. Molecules, 27(8), 2525. https://doi.org/10.3390/molecules27082525
Amira, M. B., Faize, M., Karlsson, M., Dubey, M., Frąc, M., Panek, J., Fumanal, B., Gousset-Dupont, A., Julien, J. L., Chaar, H. (2021). Fungal X-intrinsic protein aquaporin from Trichoderma atroviride: Structural and functional considerations. Biomolecules, 11(2),338. https://doi.org/10.3390/biom11020338
Athinuwat, D., Ruangwong, O. U., Harishchandra, D. L., Pitija, K., & Sunpapao, A. (2024). Biological control activities of rhizosphere fungus Trichoderma virens T1-02 in suppressing flower blight of flamingo flower (Anthurium andraeanum Lind.). Journal of Fungi, 10(1), 66. https://doi.org/10.3390/jof10010066
Boamah, S., Zhang, S., Xu, B., Zhu, N., & Li, E. (2025). Trichoderma longibrachiatum TG1 colonization and signal pathway in alleviating salinity and Fusarium pseudograminearum stress in wheat. International Journal of Molecular Sciences, 26(9),4018. https://doi.org/10.3390/ijms26094018
Carro-Huerga, G., Mayo-Prieto, S., Rodríguez-González, Á., Álvarez-García, S., Gutiérrez, S., & Casquero, P. A. (2021). The influence of temperature on the growth, sporulation, colonization, and survival of Trichoderma spp. in grapevine pruning wounds. Agronomy, 11, 1771. https://doi.org/10.3390/agronomy11091771
Ferreira, F. V., Herrmann-Andrade, A. M., Calabrese, C. D., Bello, F., Vázquez, D., & Musumeci, M. A. (2020). Effectiveness of Trichoderma strains isolated from the rhizosphere of citrus tree to control Alternaria alternata, Colletotrichum gloeosporioides and Penicillium digitatum A21 resistant to pyrimethanil in post-harvest oranges (Citrus sinensis L. (Osbeck)). Journal of Applied Microbiology, 129, 712-727. https://doi.org/10.1111/jam.14657
Ghazi Mohammed, V., Matrood, A. A. A., Rhouma, A., & Hajji-Hedfi, L. (2024). Efficacy of Beauveria bassiana and Trichoderma viride against Bemisia tabaci (Hemiptera: Aleyrodidae) on tomato plants. Journal of Biological Control, 38, 179-185. https://doi.org/10.18311/jbc/2024/36616
Hajji-Hedfi, L., Hlaoua, W., Al-Judaibi, A. A., Rhouma, A., Horrigue-Raouani, N., & Abdel-Azeem, A. M. (2023a). Comparative effectiveness of filamentous fungi in biocontrol of Meloidogyne javanica and activated defense mechanisms on tomato. Journal of Fungi, 9, 37. https://doi.org/10.3390/jof9010037
Hajji-Hedfi, L., Rhouma, A., Hajlaoui, H., Hajlaoui, F., & Rebouh, N. Y. (2023b). Understanding the influence of applying two culture filtrates to control gray mold disease (Botrytis cinerea) in tomato. Agronomy, 13, 1774. https://doi.org/10.3390/agronomy13071774
Lombardi, N., Pironti, A., Manganiello, G., Marra, R., Vinale, F., Vitale, S., Lorito, M., & Woo, S. L. (2023). Trichoderma species problematic to the commercial production of Pleurotus in Italy: Characterization, identification, and methods of control. Microbiology Research, 14(3), 1301-1318. https://doi.org/10.3390/microbiolres14030088
Matrood, A. A. A., & Rhouma, A. (2021a). Potential of the indirect and direct beneficial effects of the use of Trichoderma koningii, Aspergillus niger and Mucor sp. on eggplants plants: Plant growth and systemic resistance induction. Nova Biotechnologica et Chimica, 20, e809. https://doi.org/10.36547/nbc.809
Matrood, A. A. A., & Rhouma, A. (2021b). Evaluation of the efficiency of Paecilomyces lilacinus and Trichoderma harzianum as biological control agents against Alternaria solani causing early blight disease of eggplant. Pakistan Journal of Phytopathology, 33, 171-176. https://doi.org/10.33866/phytopathol.033.01.0673
Matrood, A. A. A., & Rhouma, A. (2022). Bioprotection of Cucumis melo from Alternaria leaf spot by Glomus mosseae and Trichoderma harzianum. Tropicultura, 40, 1-11. https://doi.org/10.25518/2295-8010.2075
Matrood, A. A. A., Rhouma, A., & Okon, O. G. (2021). Evaluation of the biological control agent’s efficiency against the causal agent of early blight of Solanum melongena. Arab Journal of Plant Protection, 39, 204-209. https://doi.org/10.22268/AJPP-039.3.204209
Matrood, A. A. A., Rhouma, A., & Mohammed, T. F. (2022). Control of Fusarium wilt disease of cucumber using rhizospheric antagonistic fungi. Arab Journal of Plant Protection, 40, 62-69. https://doi.org/10.22268/AJPP-040.1.062069
Matrood, A. A. A., Rhouma, A., Hajji-Hedfi, L., & Khrieba, M. I. (2023). Fungal diversity associated with Bemisia tabaci (Hemiptera: Aleyrodidae) on cucumber and comparative effectiveness of bioassay methods in identifying the most virulent entomopathogenic fungi. Sydowia, 75, 269-282. http://www.sydowia.at/syd75/T21-Matrood.htm
Moreno-Ruiz, D., Fuchs, A., Missbach, K., Schuhmacher, R., & Zeilinger, S. (2020). Influence of different light regimes on the mycoparasitic activity and 6-Pentyl-α-pyrone biosynthesis in two strains of Trichoderma atroviride. Pathogens, 9, 860. https://doi.org/10.3390/pathogens9100860
Othman, Y. A., Tahat, M., Alananbeh, K. M., & Al-Ajlouni, M. (2022). Arbuscular mycorrhizal fungi inoculation improves flower yield and postharvest quality component of gerbera grown under different salinity levels. Agriculture, 12, 978. https://doi.org/10.3390/agriculture12070978
Poosapati, S., Ravulapalli, P. D., Viswanathaswamy, D. K., & Kannan, M. (2021). Proteomics of two thermotolerant isolates of Trichoderma under high-temperature stress. Journal of Fungi, 7, 1002. https://doi.org/10.3390/jof7121002
Rasheela, A. R. P., Khalid, M. F., Abumaali, D. A., Alatalo, J. M., Ahmed, T. (2025). Impact of abiotic stressors on soil microbial communities: A focus on antibiotics and their interactions with emerging pollutants. Soil Systems, 9(1), 2. https://doi.org/10.3390/soilsystems9010002
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Agriculture and Environmental Science Academy

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.




